Off-label, But Lucrative
By Nav Dhillon
Big pharma profits when doctors prescribe a drug for a disorder other than the one that earned FDA approval. The practice ranges from evidence-based to highly egregious.
One in five prescriptions written in the United States is for a drug that hasn’t been approved for what’s ailing the patient.
That factoid from the Agency for Healthcare Research and Quality website casts a shadow of doubt over many of the prescriptions known as “off-label.”
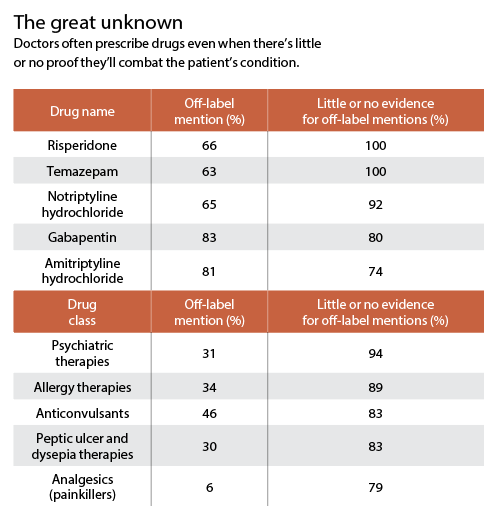
It’s when doctors prescribe a drug for a disorder other than the one that earned Food and Drug Administration (FDA) approval. They might, for example, order a diabetes drug for a patient who’s not diabetic but wants to lose weight.
Off-label prescribing is often appropriate, and...
Topics
-
Today’s the Day for Automotive Tariffs
By Ed McKinley
|The U.S. begins collecting 25% levies on imported cars today. Duties on imported car parts will follow soon. -
After Tariff-Related Selloff, Are Shares of the ‘Big 3’ Auto Stocks a Buy?
|Who’s in the trade-war driver’s seat—Ford, GM or Stellantis? -
Tired of Gold? We Could Give You Some Crypto Instead.
By Ed McKinley
|Inside Trump’s movement to create the new Fort Knox: a digital strategic reserve for Bitcoin and perhaps other tokens -
Nvidia’s Q4 Earnings: The Promise of AI Meets the Perils of Overconcentration
|The company’s AI-driven growth looks great, but does its future depend too much on data centers? Here are two takeaways from earnings. -
As Strong as Fort Knox
By Ed McKinley
|Trump and Musk want to check out one of the most secure locations in America: the United States Bullion Depository -
Letter to the Editor: Musk, Governance and America’s Future
By Luckbox
|Also, the article "Democracy in the Exponential Age," below. -
Are Robotaxis Really Driving Themselves?
By Ed McKinley
|Not quite. But with a little help from remote nannies they’re racking up impressive safety records. -
The $44 Billion Twitter ‘Catastrophe’ That Wasn’t: Musk’s Stunning Financial Turnaround at X
By Jeff Joseph
|The little-known story of how X doubled Twitter's best profits while the experts predicted "disaster” -
Waymo’s Road to IPO: Hidden Value for Alphabet Investors
|The ride-hailing company is shaking up the industry with its trailblazing autonomous driving technology -
Common Cents Dictates Some Penny-Wise Moves
By Ed McKinley
|President Trump wants to stop minting the one-cent piece, and there’s not much dissent -
Wall Street Yawns, Main Street Pays: Decoding Trump’s Trade Theater
By Ed McKinley
|Everybody talks economics when the subject of import duties comes up—that makes sense. But they’re also a powerful bargaining chip in any type of international negotiation. -
Salesforce’s New AI Push Could Transform Its Growth Trajectory
By Jeff Joseph
|The company’s ”AI agents" automate complex tasks, like answering customers’ questions -
Trends & Tickers: My Four Favorite Trade Ideas for 2025
|These are the most compelling investment themes I see for the coming year—just don’t forget the risks -
Argentina’s Economic Success: Can U.S. Learn from Milei’s Reforms?
|The U.S. faces tough fiscal choices, and Argentina’s bold reforms may provide a blueprint. -
Trump’s Panama Canal Controversy: High Tolls, Tariffs and Trade Wars
|Trump’s Panama Canal talk stirs the pot, but his proposed tariffs could really stir the markets -
Quantum Computing Is the Future—Here’s How to Invest
|From science fiction to reality—quantum computing is poised to emerge as the next big thing in tech. Why QTUM Is Your Ticket to the Future -
Will RFK Jr. Transform the Way America Makes and Sells Food?
|Politics may change the recipe for success in the food industry. Here are some potential winners and losers. -
GLP-1, the FDA and RFK-J Take on the Food Biz
By Ed McKinley
|Junk food manufacturers and restaurant owners are trying to adjust to three looming challenges -
Mark Zuckerberg 2.0: Superficial Makeover or Profound Change of Heart?
By Ed McKinley
|The billionaire CEO of Meta Platforms is shedding his dour image. We ask why. -
What Would Chris Wright’s Confirmation Mean for Nuclear Power?
By Ed McKinley
|The nominee for secretary of energy owns stock worth $40 million in an oil and gas services company





















